South Africa has made medical history by becoming the first African country and only the third worldwide to approve the twice-yearly HIV prevention injection lenacapavir (LEN), a breakthrough scientists say could help end the country’s HIV epidemic within two decades. The South African Health Products Regulatory Authority (SAHPRA) announced the approval this week, calling the drug “a game-changer” in the fight against HIV. Developed by U.S. pharmaceutical giant Gilead Sciences, lenacapavir works as a long-acting pre-exposure prophylaxis (PrEP) injection that provides almost complete protection for HIV-negative individuals.
A new era in HIV prevention
The jab, administered once every six months, prevents the virus from replicating in the body and is effective across all sexes and weight categories above 35kg. Clinical trials have shown lenacapavir to be more than 99% effective in preventing HIV infection through sexual contact. It is also considered safe for pregnant and breastfeeding women two of the most vulnerable groups in South Africa’s epidemic. “This is the most effective HIV prevention tool we’ve seen so far,” said SAHPRA CEO Dr. Boitumelo Semete-Makokotlela. “Given our high prevalence rate, lenacapavir offers real hope of turning the tide.”
Rollout and access challenges
The South African government plans to begin distribution as early as February 2026, starting with high-risk populations such as adolescent girls and young women — who account for about 1,000 new infections every week. Initial supplies will be funded through a $29.2 million Global Fund grant, enough to cover 456,000 people over two years. However, experts warn this is far below the one to two million annual doses needed to achieve nationwide impact.
Health Minister Aaron Motsoaledi said the government is working to integrate lenacapavir into the national health system and domestic financing structures. Gilead has agreed to license production to six generic drug manufacturers at a drastically reduced price of $40 per person per year, compared to the original $28,000 cost. Partnerships with the Gates Foundation and local firms are also underway to establish South African-based production in the near future.
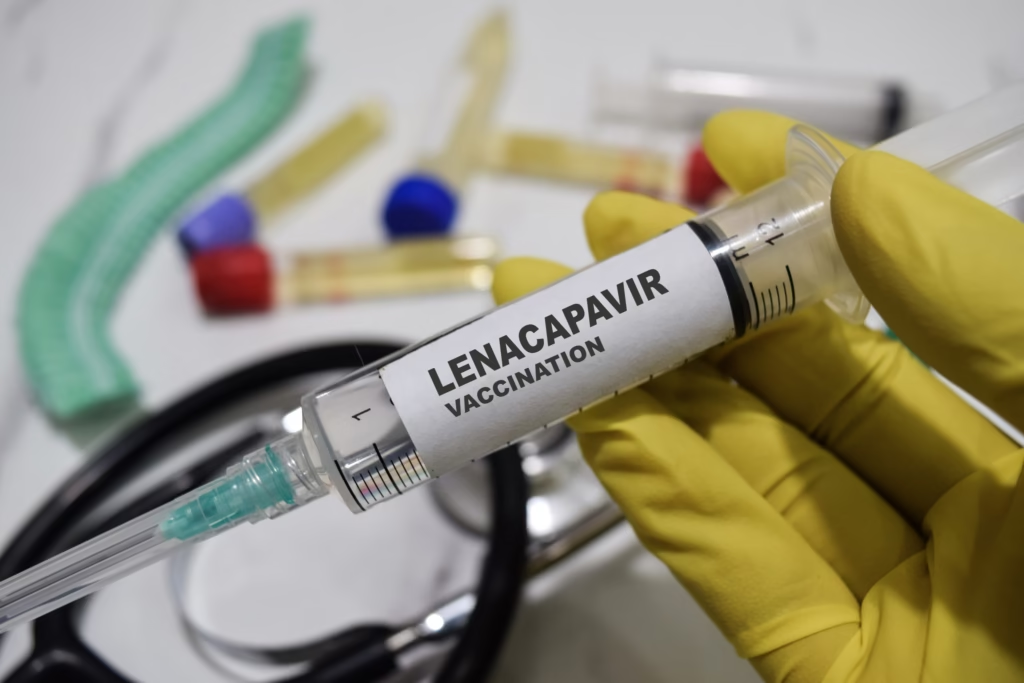
Hope for an AIDS-free generation
With roughly 8 million South Africans living with HIV — the largest number in the world the new injection could mark a turning point. If uptake is scaled up effectively, public health experts estimate South Africa could virtually eliminate HIV transmission within 14 to 18 years. Still, SAHPRA cautioned that lenacapavir should be used in combination with safe-sex practices such as condom use, to prevent other sexually transmitted infections. Motsoaledi summed up the significance: “For the first time, we have a tool powerful enough to protect our people especially our young women from HIV. The goal of an AIDS-free South Africa is finally within reach.”



